Willemite is an inorganic phosphor material used for optoelectronic applications. Much attention has been paid to the synthesis of willemite nanoparticles in the last two decades. This includes the application of new methods or modification of the existing ones. The present study proposes a polymer thermal treatment method involving calcination temperature between 500 to 1000oC to fabricate the willemite nanoparticles. The effects of synthesis parameters such as the calcination temperature, calcination holding time, PVP concentration, and the precursor concentration were extensively studied and optimized. The FT-IR, Raman and the XRD analysis revealed that the samples were amorphous at room temperature and further confirmed the formation of pure willemite nanoparticles upon the calcination process. The crystallite size of the materials ranges between 21.60-32.15 nm and increases with the increment of the calcination temperatures, calcination holding time and precursor. While the crystallite size was found to be reduced from 36.70-23.80 nm with the increase in the PVP concentration (2-5 g) for the willemite nanoparticles produced at 900oC. This is in a good agreement with the particle size determined by HR-TEM and FESEM micrographs. The Eopt values decreased with the increased of holding times over the range of 5.39 eV at 1 h to 5.27 at 4 h. The Eopt of the material was also found to be increasing from 5.24-5.32 eV with the corresponding increase in the PVP concentration. The PL emission spectra reveal a blue emission at 485 nm due to zinc interstitial. For all the synthesis condition, the PL emission was found to be depended on the particle size of the willemite. The current findings provide a pathway to reduce the high energy consumed in the synthesis of willemite nanoparticles, and the wide band gap energy of the material may have key potential applications for future optoelectronic lighting devices.
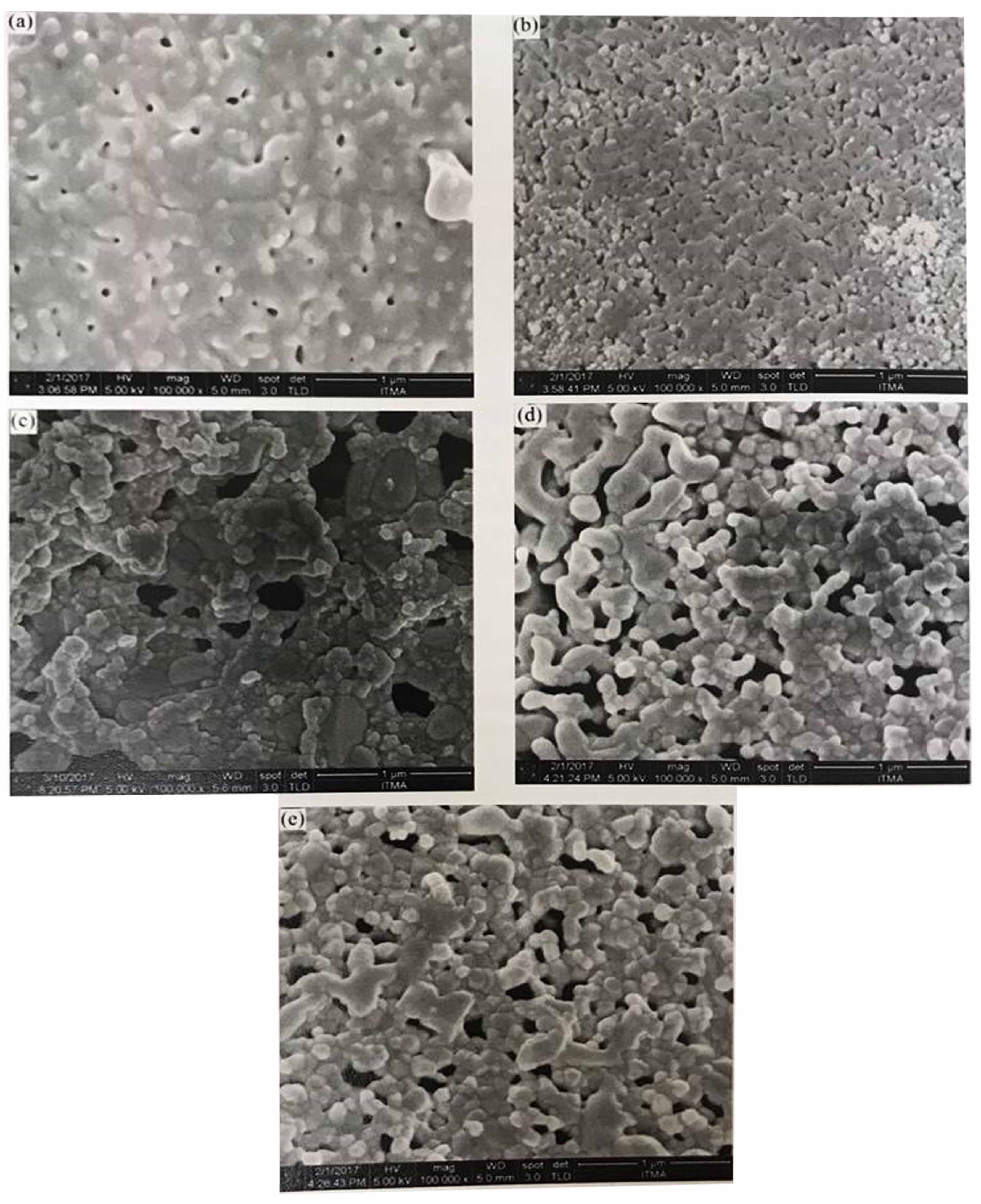
FESEM image: Nanoparticles willemite following calcination at 900°C over PVP different concentration (a) 0 g, (c) 2 g, (C), 3 g, and (c) 4 g, (d) 5g.
*Abstract of the thesis (Doctor of Philosophy) by Alibe Ibrahim Mustapha
For further information please contact:
Assoc. Prof. Dr. Khamirul Amin Matori, PhD
Chairman
khamirul@upm.edu.my
Date of Input: 25/02/2022 | Updated: 25/02/2022 | roslina_ar
MEDIA SHARING